Help us make food transparency the norm!
As a non-profit organization, we depend on your donations to continue informing consumers around the world about what they eat.
The food revolution starts with you!
Doppel Kekse Zartbittercreme - EnerBio - 330 g
Doppel Kekse Zartbittercreme - EnerBio - 330 g
This product page is not complete. You can help to complete it by editing it and adding more data from the photos we have, or by taking more photos using the app for Android or iPhone/iPad. Thank you!
×
Barra-kodea: 4305615877785 (EAN / EAN-13)
Izen arrunta: Doppelkeks mit 30% Zartbittercremefüllung
Kopurua: 330 g
Markak: EnerBio
Kategoriak: en:Snacks, en:Sweet snacks, en:Biscuits and cakes, Gaileta, en:Filled biscuits
Etiketak, ziurtagiriak, sariak:
en:Organic, en:Vegetarian, en:EU Organic, en:Non-EU Agriculture, en:Vegan, EG-Öko-Verordnung, DE-ÖKO-001, en:EU Agriculture, en:EU/non-EU Agriculture, en:Made in Germany, en:Naturland


Matching with your preferences
Health
Osagaiak
-
9 ingredients
Aleman: Weizenmehl*, Palmfett", Rübenzucker*, 9% Kakaopulver* stark entölt, 0,75 % Quinoa* gepufft und geschrotet, Weizensirup*, Backtriebmittel: Natriumhydrogencarbonat, Meersalz.Alergenoak: en:Gluten
Food processing
-
Prozesatutako jakiak
Elements that indicate the product is in the 3 - Prozesatutako elikagaiak group:
- Osagaia: Gatz arrunt
- Osagaia: Azukre
- Kategoria: Sweet snacks
Food products are classified into 4 groups according to their degree of processing:
- Prozesatu gabeko edo ahalik eta gutxien prozesatutako elikagaiak
- Sukaldaritzako osagaiak prozesatu
- Prozesatutako jakiak
- Ultra processed foods
The determination of the group is based on the category of the product and on the ingredients it contains.
Gehigarriak
-
E500
Sodium carbonate: Sodium carbonate, Na2CO3, -also known as washing soda, soda ash and soda crystals, and in the monohydrate form as crystal carbonate- is the water-soluble sodium salt of carbonic acid. It most commonly occurs as a crystalline decahydrate, which readily effloresces to form a white powder, the monohydrate. Pure sodium carbonate is a white, odorless powder that is hygroscopic -absorbs moisture from the air-. It has a strongly alkaline taste, and forms a moderately basic solution in water. Sodium carbonate is well known domestically for its everyday use as a water softener. Historically it was extracted from the ashes of plants growing in sodium-rich soils, such as vegetation from the Middle East, kelp from Scotland and seaweed from Spain. Because the ashes of these sodium-rich plants were noticeably different from ashes of timber -used to create potash-, they became known as "soda ash". It is synthetically produced in large quantities from salt -sodium chloride- and limestone by a method known as the Solvay process. The manufacture of glass is one of the most important uses of sodium carbonate. Sodium carbonate acts as a flux for silica, lowering the melting point of the mixture to something achievable without special materials. This "soda glass" is mildly water-soluble, so some calcium carbonate is added to the melt mixture to make the glass produced insoluble. This type of glass is known as soda lime glass: "soda" for the sodium carbonate and "lime" for the calcium carbonate. Soda lime glass has been the most common form of glass for centuries. Sodium carbonate is also used as a relatively strong base in various settings. For example, it is used as a pH regulator to maintain stable alkaline conditions necessary for the action of the majority of photographic film developing agents. It acts as an alkali because when dissolved in water, it dissociates into the weak acid: carbonic acid and the strong alkali: sodium hydroxide. This gives sodium carbonate in solution the ability to attack metals such as aluminium with the release of hydrogen gas.It is a common additive in swimming pools used to raise the pH which can be lowered by chlorine tablets and other additives which contain acids. In cooking, it is sometimes used in place of sodium hydroxide for lyeing, especially with German pretzels and lye rolls. These dishes are treated with a solution of an alkaline substance to change the pH of the surface of the food and improve browning. In taxidermy, sodium carbonate added to boiling water will remove flesh from the bones of animal carcasses for trophy mounting or educational display. In chemistry, it is often used as an electrolyte. Electrolytes are usually salt-based, and sodium carbonate acts as a very good conductor in the process of electrolysis. In addition, unlike chloride ions, which form chlorine gas, carbonate ions are not corrosive to the anodes. It is also used as a primary standard for acid-base titrations because it is solid and air-stable, making it easy to weigh accurately.Source: Wikipedia (Ingeles)
-
E500ii - Sodio hidrogenokarbonato
Sodium carbonate: Sodium carbonate, Na2CO3, -also known as washing soda, soda ash and soda crystals, and in the monohydrate form as crystal carbonate- is the water-soluble sodium salt of carbonic acid. It most commonly occurs as a crystalline decahydrate, which readily effloresces to form a white powder, the monohydrate. Pure sodium carbonate is a white, odorless powder that is hygroscopic -absorbs moisture from the air-. It has a strongly alkaline taste, and forms a moderately basic solution in water. Sodium carbonate is well known domestically for its everyday use as a water softener. Historically it was extracted from the ashes of plants growing in sodium-rich soils, such as vegetation from the Middle East, kelp from Scotland and seaweed from Spain. Because the ashes of these sodium-rich plants were noticeably different from ashes of timber -used to create potash-, they became known as "soda ash". It is synthetically produced in large quantities from salt -sodium chloride- and limestone by a method known as the Solvay process. The manufacture of glass is one of the most important uses of sodium carbonate. Sodium carbonate acts as a flux for silica, lowering the melting point of the mixture to something achievable without special materials. This "soda glass" is mildly water-soluble, so some calcium carbonate is added to the melt mixture to make the glass produced insoluble. This type of glass is known as soda lime glass: "soda" for the sodium carbonate and "lime" for the calcium carbonate. Soda lime glass has been the most common form of glass for centuries. Sodium carbonate is also used as a relatively strong base in various settings. For example, it is used as a pH regulator to maintain stable alkaline conditions necessary for the action of the majority of photographic film developing agents. It acts as an alkali because when dissolved in water, it dissociates into the weak acid: carbonic acid and the strong alkali: sodium hydroxide. This gives sodium carbonate in solution the ability to attack metals such as aluminium with the release of hydrogen gas.It is a common additive in swimming pools used to raise the pH which can be lowered by chlorine tablets and other additives which contain acids. In cooking, it is sometimes used in place of sodium hydroxide for lyeing, especially with German pretzels and lye rolls. These dishes are treated with a solution of an alkaline substance to change the pH of the surface of the food and improve browning. In taxidermy, sodium carbonate added to boiling water will remove flesh from the bones of animal carcasses for trophy mounting or educational display. In chemistry, it is often used as an electrolyte. Electrolytes are usually salt-based, and sodium carbonate acts as a very good conductor in the process of electrolysis. In addition, unlike chloride ions, which form chlorine gas, carbonate ions are not corrosive to the anodes. It is also used as a primary standard for acid-base titrations because it is solid and air-stable, making it easy to weigh accurately.Source: Wikipedia (Ingeles)
Ingredients analysis
-
en:Palm oil
Ingredients that contain palm oil: en:Palm fat
-
en:Vegan
No non-vegan ingredients
Unrecognized ingredients: en:Wheat syrup
-
en:Vegetarian
No non-vegetarian ingredients detected
Unrecognized ingredients: en:Wheat syrup
-
Details of the analysis of the ingredients
de: Weizenmehl, Palmfett", Rübenzucker, Kakaopulver* stark entölt 9%, Quinoa* 0.75%, Weizensirup, Backtriebmittel (Natriumhydrogencarbonat), Meersalz- Weizenmehl -> en:wheat-flour - vegan: yes - vegetarian: yes - ciqual_proxy_food_code: 9410 - percent_min: 23.395 - percent_max: 72.25
- Palmfett" -> en:palm-fat - vegan: yes - vegetarian: yes - from_palm_oil: yes - ciqual_proxy_food_code: 16129 - percent_min: 9 - percent_max: 40.625
- Rübenzucker -> en:beet-sugar - vegan: yes - vegetarian: yes - ciqual_proxy_food_code: 31016 - percent_min: 9 - percent_max: 24
- Kakaopulver* stark entölt -> en:fat-reduced-cocoa-powder - vegan: yes - vegetarian: yes - ciqual_food_code: 18100 - percent_min: 9 - percent: 9 - percent_max: 9
- Quinoa* -> en:quinoa - vegan: yes - vegetarian: yes - ciqual_food_code: 9340 - percent_min: 0.75 - percent: 0.75 - percent_max: 0.75
- Weizensirup -> en:wheat-syrup - percent_min: 0 - percent_max: 0.75
- Backtriebmittel -> en:raising-agent - percent_min: 0 - percent_max: 0.75
- Natriumhydrogencarbonat -> en:e500ii - vegan: yes - vegetarian: yes - percent_min: 0 - percent_max: 0.75
- Meersalz -> en:sea-salt - vegan: yes - vegetarian: yes - ciqual_food_code: 11082 - percent_min: 0 - percent_max: 0.73
Elikadura
-
Bad nutritional quality
⚠ ️Warning: the amount of fruits, vegetables and nuts is not specified on the label, it was estimated from the list of ingredients: 0This product is not considered a beverage for the calculation of the Nutri-Score.
Positive points: 4
- Proteinak: 4 / 5 (balioa: 7.4, rounded value: 7.4)
- Fiber: 4 / 5 (balioa: 4.5, rounded value: 4.5)
- Fruits, vegetables, nuts, and colza/walnut/olive oils: 0 / 5 (balioa: 0, rounded value: 0)
Negative points: 24
- Energia: 6 / 10 (balioa: 2089, rounded value: 2089)
- Azukreak: 5 / 10 (balioa: 24, rounded value: 24)
- Gantz saturatua: 10 / 10 (balioa: 13, rounded value: 13)
- Sodioa: 3 / 10 (balioa: 292, rounded value: 292)
The points for proteins are not counted because the negative points are greater or equal to 11.
Nutritional score: (24 - 4)
Nutri-Score:
-
Nutrient levels
-
Koipe in high quantity (25%)
What you need to know- A high consumption of fat, especially saturated fats, can raise cholesterol, which increases the risk of heart diseases.
Recommendation: Limit the consumption of fat and saturated fat- Choose products with lower fat and saturated fat content.
-
Gantz-azido ase in high quantity (13%)
What you need to know- A high consumption of fat, especially saturated fats, can raise cholesterol, which increases the risk of heart diseases.
Recommendation: Limit the consumption of fat and saturated fat- Choose products with lower fat and saturated fat content.
-
Azukre in high quantity (24%)
What you need to know- A high consumption of sugar can cause weight gain and tooth decay. It also augments the risk of type 2 diabetes and cardio-vascular diseases.
Recommendation: Limit the consumption of sugar and sugary drinks- Sugary drinks (such as sodas, fruit beverages, and fruit juices and nectars) should be limited as much as possible (no more than 1 glass a day).
- Choose products with lower sugar content and reduce the consumption of products with added sugars.
-
Gatz arrunt in moderate quantity (0.73%)
What you need to know- A high consumption of salt (or sodium) can cause raised blood pressure, which can increase the risk of heart disease and stroke.
- Many people who have high blood pressure do not know it, as there are often no symptoms.
- Most people consume too much salt (on average 9 to 12 grams per day), around twice the recommended maximum level of intake.
Recommendation: Limit the consumption of salt and salted food- Reduce the quantity of salt used when cooking, and don't salt again at the table.
- Limit the consumption of salty snacks and choose products with lower salt content.
-
-
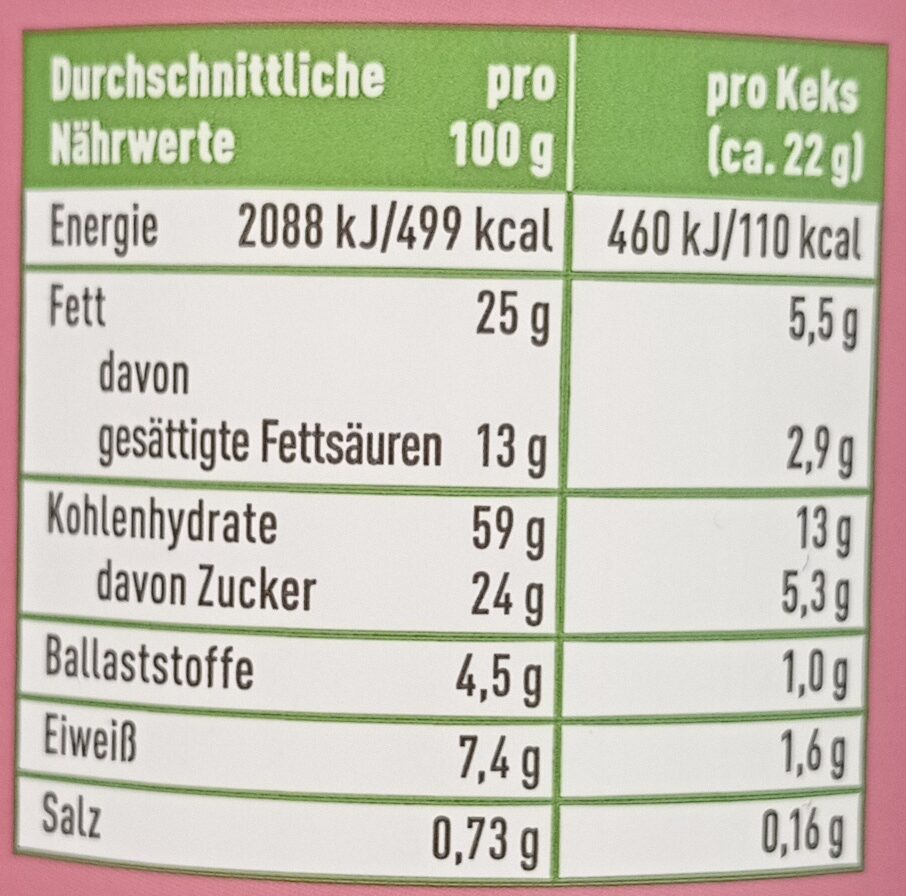
Nutrition facts
Nutrition facts As sold
for 100 g / 100 mlAs sold
per serving (22g)Compared to: en:Filled biscuits Energia 2.089 kj
(499 kcal)460 kj
(110 kcal)+% 4 Koipe 25 g 5,5 g +% 23 Gantz-azido ase 13 g 2,86 g +% 41 Carbohydrates 59 g 13 g -% 12 Azukre 24 g 5,28 g -% 25 Fiber 4,5 g 0,99 g +% 54 Proteina 7,4 g 1,63 g +% 33 Gatz arrunt 0,73 g 0,161 g +% 21 Fruits‚ vegetables‚ nuts and rapeseed‚ walnut and olive oils (estimate from ingredients list analysis) 0 % 0 %
Ingurumena
-
Eco-Score C - Moderate environmental impact
The Eco-Score is an experimental score that summarizes the environmental impacts of food products.→ The Eco-Score was initially developped for France and it is being extended to other European countries. The Eco-Score formula is subject to change as it is regularly improved to make it more precise and better suited to each country.Life cycle analysis
-
Average impact of products of the same category: B (Score: 70/100)
Kategoria: Biscuit (cookie)
Kategoria: Biscuit (cookie)
- PEF environmental score: 0.34 (the lower the score, the lower the impact)
- including impact on climate change: 2.86 kg CO2 eq/kg of product
Stage Impact Agriculture
80.2 %Processing
12.0 %Ontziratzea
3.2 %Transportation
3.2 %Distribution
1.4 %Consumption
0.0 %
Bonuses and maluses
-
Labels with high environmental benefits
Bonus: +15
-
EU Organic
Organic agriculture contributes to preserve biodiversity, climate, water quality and soil fertility.
Organic food is food produced by methods complying with the standards of organic farming and features practices that cycle resources, promote ecological balance, and conserve biodiversity.
-
-
Missing origins of ingredients information
Malus: -5
⚠ ️ The origins of the ingredients of this product are not indicated.
If they are indicated on the packaging, you can modify the product sheet and add them.
If you are the manufacturer of this product, you can send us the information with our free platform for producers.
-
Ingredients that threatens species
Malus: -10
Contains palm oil
Tropical forests in Asia, Africa and Latin America are destroyed to create and expand oil palm tree plantations. The deforestation contributes to climate change, and it endangers species such as the orangutan, the pigmy elephant and the Sumatran rhino.
-
Missing packaging information for this product
Malus: -15
⚠ ️ The information about the packaging of this product is not filled in.⚠ ️ For a more precise calculation of the Eco-Score, you can modify the product page and add them.
If you are the manufacturer of this product, you can send us the information with our free platform for producers.
Eco-Score for this product
-
Impact for this product: C (Score: 55/100)
Produktua: Doppel Kekse Zartbittercreme - EnerBio - 330 g
Life cycle analysis score: 70
Sum of bonuses and maluses: -15
Final score: 55/100
-
Carbon footprint
-
Equal to driving 1.5 km in a petrol car
286 g CO² per 100g of product
The carbon emission figure comes from ADEME's Agribalyse database, for the category: Biscuit (cookie) (Source: ADEME Agribalyse Database)
Stage Impact Agriculture
82.7 %Processing
8.0 %Ontziratzea
3.8 %Transportation
4.7 %Distribution
0.7 %Consumption
0.0 %
Ontziratzea
-
Missing packaging information for this product
⚠ ️ The information about the packaging of this product is not filled in.Take a photo of the recycling information Take a photo of the recycling information
Transportation
-
Origins of ingredients
Missing origins of ingredients information
⚠ ️ The origins of the ingredients of this product are not indicated.
If they are indicated on the packaging, you can modify the product sheet and add them.
If you are the manufacturer of this product, you can send us the information with our free platform for producers.Add the origins of ingredients for this product Add the origins of ingredients for this product
Threatened species
-
Contains palm oil
Drives deforestation and threatens species such as the orangutan
Tropical forests in Asia, Africa and Latin America are destroyed to create and expand oil palm tree plantations. The deforestation contributes to climate change, and it endangers species such as the orangutan, the pigmy elephant and the Sumatran rhino.
Etiketak
-
EU Organic
Organic agriculture contributes to preserve biodiversity, climate, water quality and soil fertility.
Organic food is food produced by methods complying with the standards of organic farming and features practices that cycle resources, promote ecological balance, and conserve biodiversity.
Report a problem
-
Incomplete or incorrect information?
Category, labels, ingredients, allergens, nutritional information, photos etc.
If the information does not match the information on the packaging, please complete or correct it. Open Food Facts is a collaborative database, and every contribution is useful for all.
Datuen iturria
Product added on by kiliweb
Last edit of product page on by femmenoire.
Produktuaren orria -gatik editatua fabi2, moon-rabbit, roboto-app, segundo, yuka.sY2b0xO6T85zoF3NwEKvlkliAub0pBjhFgXfg1eq286WB6WyfMNC8JihCas.